Description
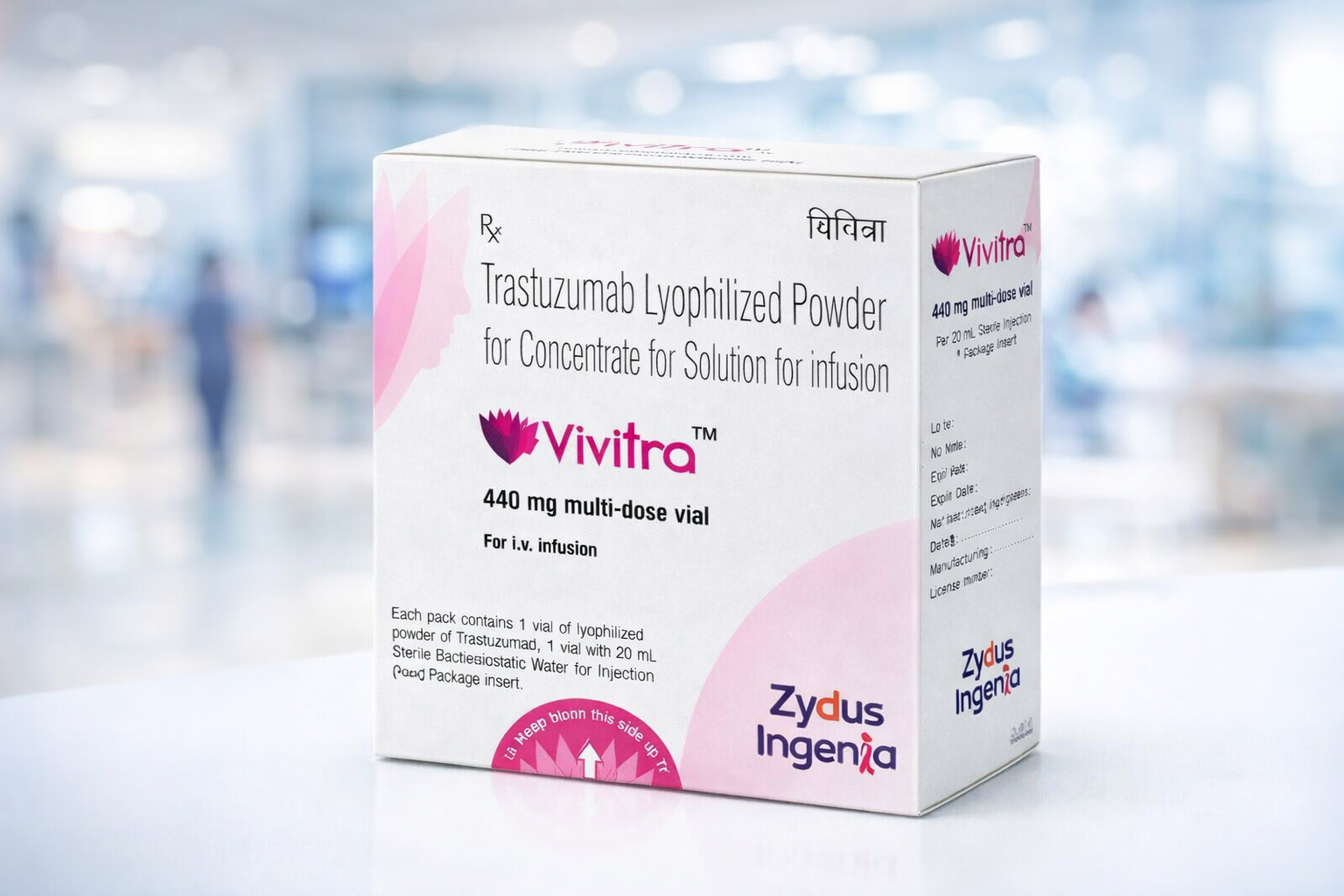
Vivitra 440 mg contains Trastuzumab, a recombinant humanized monoclonal antibody directed against the human epidermal growth factor receptor 2 (HER2) cancer. It is supplied as a lyophilized (freeze-dried) powder that is reconstituted and administered intravenously in a clinical setting.
🔬 Mechanism of Action
Trastuzumab binds selectively to the HER2 receptor on cancer cells. As a result, it:
- Inhibits HER2-mediated cell proliferation
- Activates antibody-dependent cellular cytotoxicity (ADCC)
- Prevents tumor growth and survival signaling
This targeted approach makes it highly effective in HER2-overexpressing cancers.
💊 Indications
Vivitra 440 mg is indicated for:
- HER2-positive early breast cancer (adjuvant and neoadjuvant settings)
- HER2-positive metastatic breast cancer
- HER2-positive metastatic gastric or gastroesophageal junction cancer
It is often used in combination with chemotherapy or other targeted therapies.
📌 Key Features
- Strength: 440 mg
- Dosage Form: Lyophilized powder for intravenous infusion
- Therapeutic Class: Anti-HER2 Monoclonal Antibody
- Administration: IV infusion under oncologist supervision
- Monitoring: Cardiac function monitoring (e.g., LVEF assessment) required during therapy
⚕️ Benefits
- Targeted HER2 blockade
- Improves progression-free and overall survival
- Standard of care in HER2-positive breast cancer
- Can be used across early and metastatic stages
⚠️ Precautions
- Risk of cardiotoxicity; regular cardiac monitoring required
- Infusion-related reactions may occur
- Not recommended during pregnancy
- Use strictly under oncology supervision
Vivitra 440 mg (Trastuzumab Lyophilized Powder) is a cornerstone targeted therapy in HER2-positive cancers, offering improved survival and disease control when administered as part of a comprehensive oncology treatment plan.